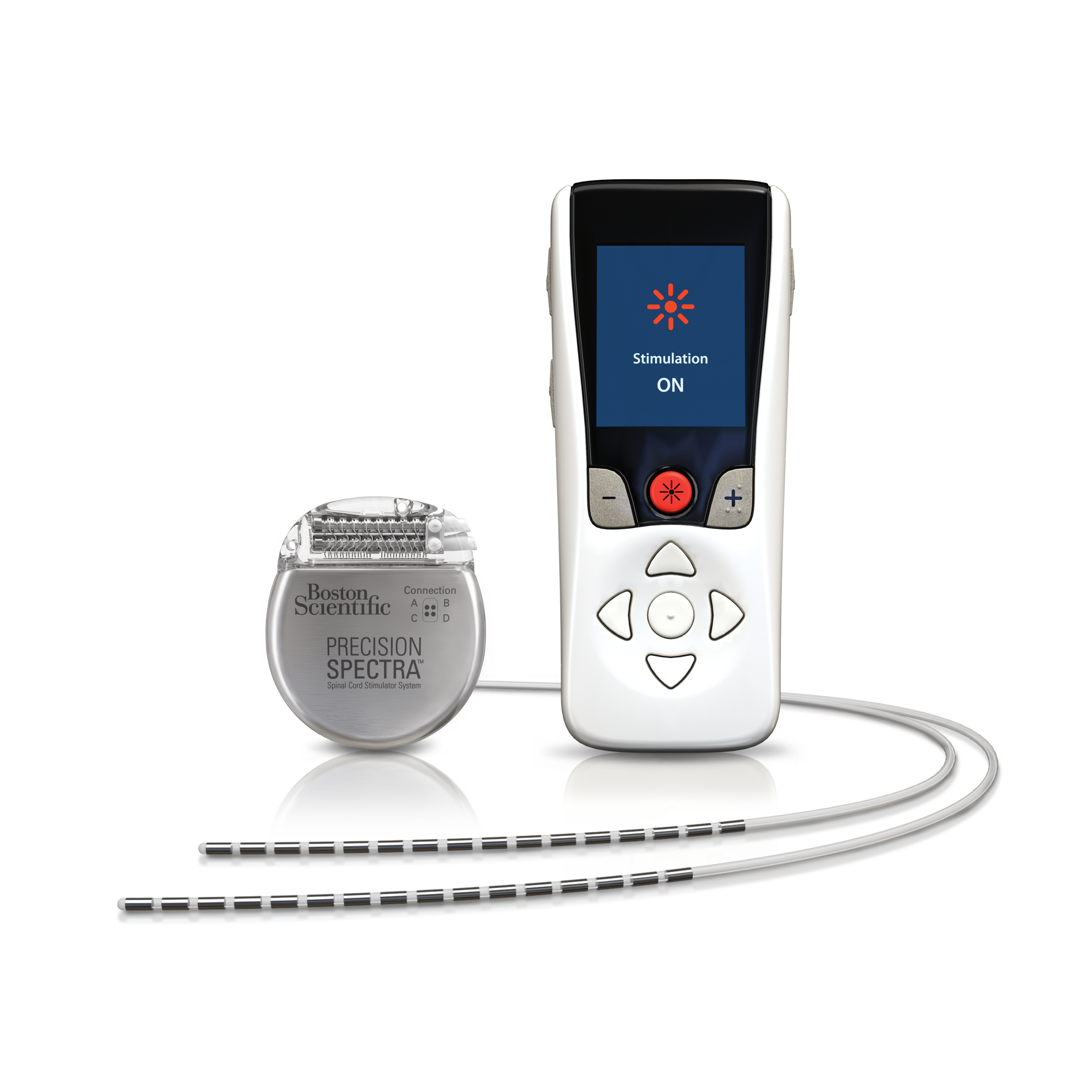 
What will it accomplish? The SCS system may help treat chronic, intractable pain in a patient’s trunk or limbs and pain associated with diabetic neuropathy. For questions about this issue or to report patients who are unable to exit MRI mode, please contact your local Abbott representative or Abbott Technical Support at 1-80 (option 3) for. When is it used? The SCS system is used as an aid to manage chronic pain in the trunk or limbs, including one-sided or two-sided pain associated with failed back surgery syndrome, intractable low back pain, leg pain, and pain associated with nerve damage caused by diabetes ( diabetic neuropathy). The Infinity Deep Brain Stimulation system delivers mild pulses of electricity to the precise areas within the brain. 24, 2022 /PRNewswire/ - Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it has received U.S. The external remote control is battery operated and can be controlled by the patient or a health care provider. The Proclaim DRG Neurostimulation System is the first and only neurostimulation device approved only for complex regional pain syndrome (CRPS) and those seeking relief from causalgia -nerve pain following surgery or injury. The latest regulatory nod covers all of the spinal cord stimulators that Abbott currently offers in the U.S. The US Food and Drug Administration (FDA) has approved new expanded MRI compatibility for Abbott’s Proclaim XR Spinal Cord Stimulation (SCS) System with Octrode leads. How does it work? The implanted signal generator receives radio signals from the remote control which tell the signal generator when to deliver appropriate stimulation to the spinal cord. This supplement expands the Senza SCS systems’ indications for use to include lower limb pain associated with diabetic neuropathy, also known as painful diabetic neuropathy. The main components of the SCS system include an implanted signal generator that is connected to one or two implanted leads and a remote control that can turn the stimulator ON or OFF to allow adjustments of therapy settings. What is it? The Senza Spinal Cord Stimulation (SCS) system is an implanted, rechargeable spinal cord stimulation system intended to treat long-term (chronic) pain in the trunk or limbs that is difficult to manage (intractable). Product Name: Senza Spinal Cord Stimulation (SCS) SystemĪddress: 1800 Bridge Parkway, Redwood City, CA 94065 See the links below to the Summary of Safety and Effectiveness Data and product labeling for more information on this product, its indications for use, and the basis for the FDA’s approval. This is a brief overview of information related to the FDA’s approval to market this product.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
